- No products in the cart.
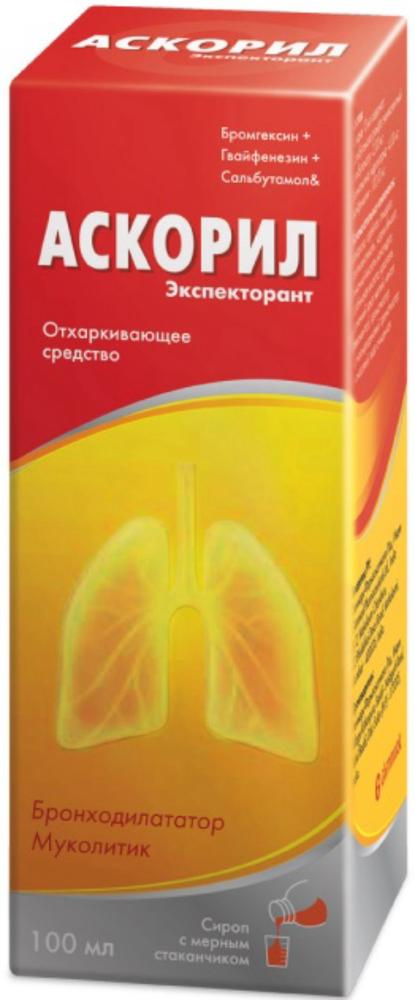
Ascoril expectorant syrup 100ml vials
$6.00
Ascoril expectorant syrup 100ml vials
Description
Composition
Active substance:
10 ml of syrup contains: salbutamol sulfate, salbutamol equivalent – 2.00 mg, bromhexine hydrochloride – 4,00 mg, guaifenesin – 100.00 mg ;.
Excipients:
Sucrose (sugar), sorbitol (70% solution), glycerol (glycerine), propylene glycol, sodium benzoate, citric acid monohydrate, sorbic acid, colorant sunset yellow (FCF), menthol (levomenthol), flavor blackcurrant (perfume blackcurrant ID 20158 ), flavor pineapple (pineapple flavor super “PH”), purified water.
Description:
A clear, orange-colored, viscous liquid with a characteristic odor.
Product form:
Syrup 4 mg + 100 mg + 2 mg / 10 ml plastic vials dark.
Bottles of 100 ml or 200 ml.
Contraindications
Hypersensitivity, tachyarrhythmia, myocarditis, aortic stenosis, decompensated diabetes, thyrotoxicosis, glaucoma, hepatic and / or renal failure; gastric ulcer and duodenum 12 (in the acute stage), hypertension, gastric bleeding, pregnancy, lactation.
Carefully:
Diabetes, hypertension, peptic ulcer and duodenal ulcer in remission.
Dosage
2 mg + 50 mg + 1 mg / 5 ml
Indications
In the combined therapy of acute and chronic bronchopulmonary diseases accompanied by formation of difficult to separate viscous secretion: bronchial asthma; tracheobronchitis; obstructive bronchitis; pneumonia; emphysema; whooping cough; pneumoconiosis; pulmonary tuberculosis and others.
Interaction with other drugs
Other beta2-Adrenomimeticalkie funds and theophylline enhance the effect of salbutamol and increase the likelihood of side effects.
Ascoril expectorant not administered with drugs containing codeine and other antitussive, as this makes it difficult expectoration of liquefied sputum.
Part of the drug Ascoril expectorant bromhexine promotes penetration of antibiotics (erythromycin, cephalexin, oxytetracycline) in pulmonary tissue.
Do not use the drug Ascoril expectorant simultaneously with non-selective beta-adrenoceptor blockers, such as propranolol.
Part of the drug salbutamol Ascoril expectorant is not recommended for patients who receive inhibitors of monoamine oxidase (MAO).
Diuretics and drugs glucocorticoids increase hypokalemic effect of salbutamol.
Do not take simultaneously with Ascoril expectorant alkaline water.
Overdose
Symptoms: increasing incidence of side effects, except dozonezavisimyh allergic reactions.
Treatment: symptomatic therapy.
pharmachologic effect
Pharmacological group:
Expectorant drugs.
Pharmacological properties:
Expectorant combination.
Pharmacodynamics:
Combined preparation has a bronchodilator, an expectorant and mucolytic action.
Salbutamol – a bronchodilator and stimulates beta2-adrenergic receptors of the bronchi, blood vessels and myometrium. Prevents or eliminates bronchospasm, reducing airway resistance increases lung capacity. Causes expansion of the coronary arteries, does not lower blood pressure.
Bromhexinum – mucolytic agent. Has expectorant action, improves expectoration.
Guaifenesin – mucolytic agent reduces the surface tension structures bronchopulmonary apparatus; stimulates secretory bronchial mucosa cells that produce neutral polysaccharides depolymerizes acidic mucopolysaccharides, reduces the viscosity of sputum, facilitates the removal of sputum and facilitates the transition nonproductive cough productive.
Pharmacokinetics:
Salbutamol: ingestion absorption – high. Food intake reduces the rate of absorption, but does not affect the bioavailability. Relationship to plasma proteins – 10%. It crosses the placenta.
Undergoes first-pass metabolism in the liver and in the intestinal wall by fenolsulfotransferazy inactivated to 4-O-sulfate ester. The half-life (T1 / 2) -. 3.8-6 hours excreted by the kidneys (69-90%), preferably in the form fenolsulfatnogo inactive metabolite (60%) for 72 hours and in the bile (4%). Bioavailability of orally administered salbutamol is about 50%.
Bromhexine: When administered almost completely (99%) absorbed in the gastrointestinal tract (GIT) for 30 min. Bioavailability – low (primary effect “pass” through the liver). It crosses the placental and blood-brain barriers. The liver is demethylation and oxidation, is metabolised to a pharmacologically active ambroxol. T1 / 2 -15 hr (due to slow reverse diffusion from the tissue). Excreted by the kidneys. In chronic renal failure disturbed the excretion of metabolites. Repeated application may accumulate.
Gvayfenezin: absorption from the gastrointestinal tract – fast (25-30 minutes after ingestion). T1 / 2 – 1 h. Penetrates into the tissue containing acidic mucopolysaccharides.
Approximately 60% of the administered drug is metabolized in the liver. Light output (sputum) and kidneys in unchanged form and as inactive metabolites.
Pregnancy and breast-feeding
The drug is contraindicated during pregnancy and lactation.
Conditions of supply of pharmacies
On prescription.
side effects
Rare: when used in high doses can sometimes occur headache, dizziness, irritability, sleep disturbance, drowsiness, tremors, convulsions, nausea, vomiting, diarrhea, acute gastric ulcer and duodenal ulcers, heart palpitations, perhaps urine staining pink , allergic reactions (rash, urticaria), collapse, bronchospasm.
special instructions
Guaifenesin urine stains pink.
It is not recommended to take the drug at the same time alkaline water.
Storage conditions
In a dry, dark place at a temperature not higher than 25 C.
Keep out of the reach of children.
Dosing and Administration
Inside. Adults and children over 12 years of 10 ml (2 teaspoons) 3 times / day.
Children under the age of 6 – 5 ml (1 teaspoon) 3 times / day, 6 to 12 years – 5-10 ml (1-2 teaspoons) 3 times / day.
Information
Appearance may differ from that depicted in the picture. There are contraindications. You need to read the manual or consult with a specialist
Additional information
| Weight | 0.100 kg |
|---|---|
| Manufacturer | Glenmark |










There are no reviews yet.